Australian researchers have found that zinc can ‘starve’ one of the world’s most deadly microbes by preventing its uptake of an essential metal.
The finding, by infectious disease researchers at the University of Adelaide and The University of Queensland (UQ), opens the way for further work to design antibacterial agents in the fight against Streptococcus pneumoniae.
These bacteria are responsible for more than a million deaths a year, killing children, the elderly and other vulnerable people by causing pneumonia, meningitis and other serious infectious diseases.
Project leader Dr Christopher McDevitt, from the University of Adelaide’s Research Centre for Infectious Diseases, said the study found that zinc “jammed shut” a protein transporter in the bacteria so it could not take up manganese.
Manganese is an essential metal that Streptococcus pneumoniae needs to invade and kill humans.
“It’s long been known that zinc plays an important role in the body’s ability to protect against bacterial infection, but this is the first time anyone has been able to show how zinc actually blocks an essential pathway, causing the bacteria to starve,” Dr McDevitt said.
Professor Bostjan Kobe from UQ’s School of Chemistry and Molecular Biosciences said the work spanned fields from chemistry and biochemistry to microbiology and immunology.
“We can now see, at an atomic level of detail, how this transport protein is responsible for keeping the bacteria alive by scavenging one essential metal (manganese), but at the same time also makes the bacteria vulnerable to being killed by another metal (zinc)," Professor Kobe said.
Professor Matt Cooper from UQ’s Institute for Molecular Bioscience (IMB) said antibiotic-resistant strains of Streptococcus pneumoniae emerged more than 30 years ago, with up to 30 per cent of these bacterial infections now considered multi-drug resistant.
“The Centers for Disease Control classify multi-drug resistant Streptococcus pneumoniae as a serious threat, with more than one million cases per year in the US alone,” Professor Cooper said.
“New treatments are urgently needed and our research has provided insights into how the uptake of metal ions affects the ability of Streptococcus pneumoniae to cause disease.”
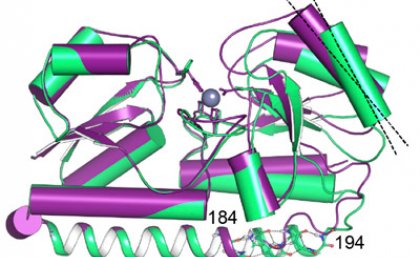
The study reveals that the bacterial transporter (PsaBCA) uses a ‘spring-hammer’ mechanism that binds zinc and manganese in different ways because of their difference in size.
The smaller size of zinc means that when it binds to the transporter, the mechanism closes too tightly around the zinc, causing an essential spring in the protein to unwind too far, jamming it shut and blocking the transporter from being able to take up manganese.
Dr McDevitt said without manganese, the immune system could easily clear the body of these bacteria.
“For the first time, we understand how these types of transporters function,” he said.
“With this new information we can start to design the next generation of antibacterial agents to target and block these essential transporters.”
The research, funded by the Australian Research Council and the National Health and Medical Research Council, has been published in the scientific journal Nature Chemical Biology.
Contact Dr Mark Ashton (m.ashton@uniquest.com.au) from UQ commercialisation arm Uniquest Pty Limited to discuss commercial opportunities around this research..
Situated within the Faculty of Science, the School of Chemistry and Molecular Biosciences teaches and researches in the disciplines of Chemistry, Biochemistry, Biotechnology, Microbiology and Parasitology.
The Institute for Molecular Bioscience (IMB) is a research institute of The University of Queensland that aims to improve quality of life by advancing medical genomics, drug discovery and biotechnology.